The ultraviolet catastrophe is a problem in physics arising from the consideration of black body radiation. A black body absorbs and re - emits all the electromagnetic radiation falling on it. As the object heats up the average frequency and energy of the radiation given off increase.
A problem arose when calculating the power radiated by a black body. The best model available was then the simple harmonic oscillator and this said the number of modes of oscillation in a 3D box was proportional to the square of the frequency of the wave and that the power of a wave was proportional to the frequency of it. The equipartition theorem said each mode of vibration was a degree of freedom, and that it had a fixed amount of energy, proportional to the temperature. As you increase the number of vibration modes, you get a much bigger increase in the the number of degrees of freedom and energy. In fact the energy increases to infinity according to classical theory. In fact any object above absolute zero should emit an infinite amount of radiation, most of it as the highest energy shortest wavelength rays.
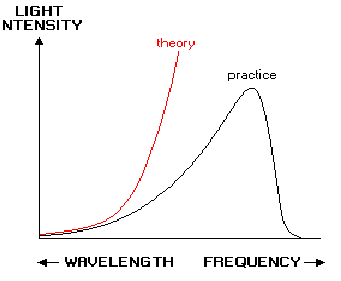
Planck modified the classical harmonic oscillator model for radiation by introducing quantized emission. Radiation was produced as discrete packets of energy called photons. These packets have energy proportional to their frequency (E=hf, where![]() is called Planck's constant). This reduced the number of available modes in the cavity for higher frequencies, causing the power to tend to zero at high frequencies.
is called Planck's constant). This reduced the number of available modes in the cavity for higher frequencies, causing the power to tend to zero at high frequencies.
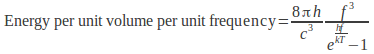
Now the intensity did not go to infinity and the power found from Planck's equation agreed with experiment.