Conduction is that process of heat transfer which occurs without any overall movement of the substance. Instead, heating one part of a material gives the atoms, molecules and any electrons free to move kinetic energy. They start to vibrate more energetically – heat energy is equivalent to random kinetic energy of all types, rotational, translational and vibrational - hitting surrounding atoms, molecules and electrons, causing them to vibrate. In this way heat energy is passed along, or conducted along a material.
Not all materials conduct well. The most efficient conductors are metals. Metals contain electrons free to move throughout the lattice of the metal. Electrons are light and because they are free to move throughout the metal, may be given a wide range of energy. Most materials that do not contain free electrons are bad conductors. The only way heat can be transmitted from one part to another is by the vibrations of atoms and molecules exciting neighbouring atoms and molecules. Wood is a bad conductor as are most gases and liquids.
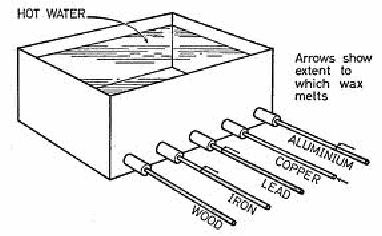
The diagram below shows how good conductors transmit heat along the length of a rod of the material so if covered in wax and placed in contact with hot water, the good conductors will conduct heat allowing much of the wax to melt while bad conductors will not.
Diamond is a counterexample. It has no electrons free to move but is a very good conductor of heat, because of it's outstanding hardness. Diamond is the hardest material known. The hardness of diamond means that the vibration of any part is rapidly transmitted to any other part of the diamond structure.