After the invention of the simple cell, with it's associated problem of polarization (bubbles of hydrogen forming at the positive carbon electrode, increasing the resistance of the cell and reducing the current), a number of different designs of cell came along in which polarization was either reduced or eliminated. One of the most successful was the Leclanche cell, invented in 1865. It has an EMF of about 1.5 V and was used for electric bells and telegraphs.
The positive pole is a carbon rod surrounded by a depolarizing mixture of powdered and manganese oxide in a porous pot. This pot is placed in a glass jar containing an ammonium chloride solution. A zinc rod is placed in the ammonium chloride solution.
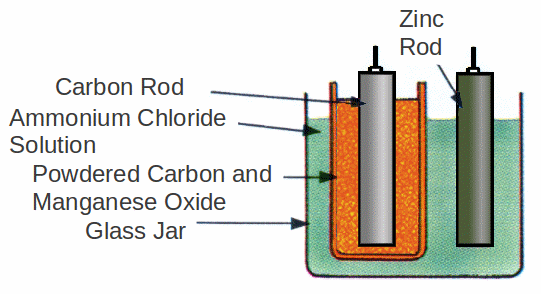
Polarization is prevented by the manganese oxide with oxidises the hydrogen to form water. This process is slow however so that a large current cannot be supplied for long without the cell 'resting' so that all the hydrogen produced can be oxidised. This makes the cell suitable only for intermittent use in bells or or telegraphs. Occasionally water must be added to make up for evaporation losses otherwise these cells will run for a long time without attention.