An ideal gas is only ideal within certain limits of temperature, pressure and volume. Within these limits the behaviour of the gas will be described by
Boyle's Law:![]() if the temperature is kept constant.
if the temperature is kept constant.
Charles's Law:![]() if the pressure is kept constant.
if the pressure is kept constant.
The Pressure Law:![]() if the volume is kept constant.
if the volume is kept constant.
These can be combined into the Ideal Gas Equation, or The Equation of State:![]() (1) where
(1) where
p = Pressure in Pa or N/m 2
V = Volume in m 3
n = number of moles in the gas mixture.
R = 8.314 J/mol K. - The Universal Gas Constant or Universal Molar Gas Constant.
T = Temperature.
No reference is made in the above equation to the type of gas or the mass or type of molecules – whether large or small.. In fact these factors are irrelevant – the equations apply to all type of gas withou modification. In fact it is part of the theory that all gases at s.t.p. - standard temperature (0 degrees Celsius, 273.15 degrees Kelvin) and pressure (1.013*10 5 Pa) occupy the same volume of 22400 cm 3.
The equation![]() (1) is not the only possible form of the equation of state. We may express it in terms of the average kinetic energy of the gas molecules:
(1) is not the only possible form of the equation of state. We may express it in terms of the average kinetic energy of the gas molecules:
![]() (2)
(2)
The kinetic energy of a particle mass![]() is
is![]() Together with the equation above we have
Together with the equation above we have![]() where N is the total number of gas molecules.
where N is the total number of gas molecules.
By putting the right hand side of (1) equal to the right hand side of (2) we obtain
![]() so
so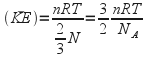
n is the number of moles and N is the number of atoms so![]() Avagadro's Number (=6.023*10 23 ).
Avagadro's Number (=6.023*10 23 ).
Hence![]() where k=Boltsmann's constant =1.38*10 -23 J/molecule K
where k=Boltsmann's constant =1.38*10 -23 J/molecule K
