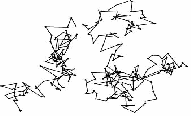A British botanist Robert Brown, was observing through a microscope some very tiny particles of pollen in suspension in water. He he saw that some of the smaller particles were moving about continuously in a haphazard way. It is due to the bombardment of tiny particles by the liquid molecules. The direction of the resultant force is constantly changing as at different times the pollen grain is hit more on one side than another, hence the random nature of the motion. Thus 'Brownian motion' is defined as the random or zig zag motion of the suspended molecules.
Experiment :
Brownian motion can be demonstrated by releasing some smoke particles from burning cord into a small glass container and putting a cover plate to seal the smoke and air into the cell.
To see brownian motion in a liquid place some water with graphite particles suspended in it into the container.
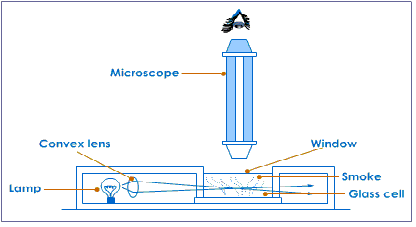
Now adjust the microscope slightly until you can see very bright graphite or smoke particles. The particles of graphite (or smoke) scatter (reflect) the light shining on them and so appear as bright points of light darting about in a random or erratic motion. Note that the graphite (or smoke) particles are much larger than the water (or air) molecules. The particles can be seen by the light they scatter but the molecules themselves are too small to be seen.
The irregular movement of the visible particles of graphite (or smoke) is explained as being due to an uneven bombardment of the particles by the invisible molecules of water (or air). The irregular path followed by an individual smoke or graphite particle is shown below.