There are usually said to be three types of nuclear radiation - alpha, beta and gamma radiation.
Alpha radiation consists of alpha particles. An alpha particle consists of two protons and two neutrons. It is a helium nucleus.
Beta radiation consists of fast or high energy electron. When a nucleus undergoes beta decay, one of the neutrons in the nucleus becomes a proton, and a particle called anti – neutrino is also emitted. In alpha and beta decay the atom changes and becomes another element. When a nucleus undergoes gamma decay, the element does not change. The nucleus just becomes less energetic.
All nuclear radiation is emitted when the nucleus undergoes some sort of decay. Alpha or beta decay the mean the atom changes from one element into another:
Alpha Decay:![]()
Beta Decay:![]()
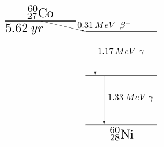
Gamma decay:![]() Sometimes an element may undergo a series of decays, called a decay series The
Sometimes an element may undergo a series of decays, called a decay series The
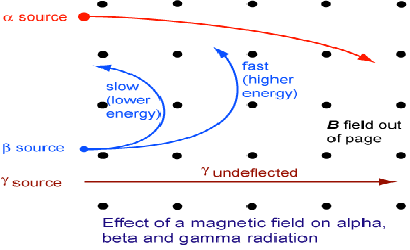
The three types of radiation can be told apart using magnetic fields. Gamma radiation is not charged, hence it will not be affected by the field and will pass through travelling in a straight line. Alpha radiation is positively charged and beta radiation is negatively charged and they will curl in opposite directions as predicted by the left hand rule.
Radioactivity is dangerous and can cause radiation sickness and kill.
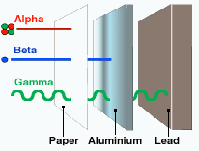
Alpha radiation can be stopped very easily with a piece of paper or a few cm of air, but if it is swallowed it is the most deadly because all the radiation is absorbed by the body. Beta radiation can be stopped with a few cm of aluminium and gamma radiation is the most penetrating and needs several cm of lead. Gamma radiation is the least dangerous is a source of gamma radiation is swallowed because most of it escapes.
The three types of radiation have their uses. Alpha radiation is used in smoke detectors, beta radiation is used to control the thickness of sheet metal and gamma rays are used to treat cancer.
