From chemical studies we know that molecules of oils and fats are longer than they are wide and that one end is attracted to water, If a drop of oil is placed on a water surface it floats and spreads out to form a thin layer with the molecules standing upright like a piece of carpet pile. When the spreading stops it is reasonable to assume that the layer is one molecule thick.
A suitable substance is oleic acid prepared as a 0.5% solution in![]() of methanol. When this is added to water the methanol partly evaporates and partly dissolves in the water leaving a film of oleic acid.
of methanol. When this is added to water the methanol partly evaporates and partly dissolves in the water leaving a film of oleic acid.
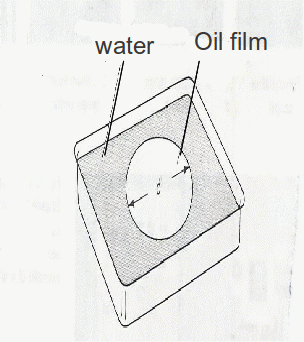
Assuming the film to be a flat cylinder of thickness![]() where
where![]() is the length of a molecule then
is the length of a molecule then
![]()
Hence the length of a molecule![]()
Typically![]()
