There are three methods of heat transfer: Conduction, Convection and Radiation.Regardless of the method, heat always travels from hot to cold.
Conduction
This is the method of heat transfer in solids.
It occurs very slightly in liquids and gases. It is a slow process compared to convection and radiation and is the slowest of the three.
Good conductors - all metals. Diamond.
Bad conductors - air, wood, cork, wool, fur, feathers, plastic, polystyrene, fibreglass, glass, fabrics, foam.
Comparing metals
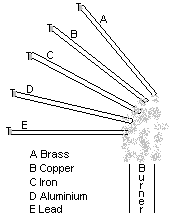
The heat from the flame is conducted along the length of each metal rod. Metals do not all conduct heat at the same rate - some are better conductors than others. When the heat reaches the paraffin wax it stars to melt and eventually the pin falls off. The rod whose pin falls off first is the best conductor. In order of conductivity they are:
Copper > aluminium > brass > iron > Lead
Comparing conductors and insulators

When heated in the middle by passing it through a flame several times, the paper on the brass side is scorched but that on the wood side is badly scorched. This is because the brass, being a good conductor, conducts the heat away quickly preventing scorching of the paper on that side.
Uses of conductors and insulators
If heat is required to travel quickly through a solid then a good conductor is used. Kettles, saucepans boilers radiators are made of metals.
If heat is not required to travel quickly then an insulator (bad conductor) is used e.g. the handles of saucepans, kettles etc., are made of substances such as wood or plastic.
Materials that trap air are very good insulators, since air is such a bad conductor. Wool, fur, feathers, fibreglass contain tiny pockets of trapped air which reduce conduction making them good insulators.
Air on its own is also used as an insulator. Layers of still air are used because they are such bad conductors e.g. air in the cavity between walls and in double glazing help to reduce heat loss by conduction.
Conduction in Non-metals
When a material heats up, the atoms gain more kinetic energy (random vibrations equate to heat energy) and vibrate more rapidly. IN a sold, the atoms will vibrate about a fixed position, jostling surrounding atoms, causing them to vibrate and so transferring kinetic energy to them. These in turn jostle neighbouring atoms and so cause energy to be transferred throughout the material.
Conduction in Metals
The above, slower process also occurs in metals. In metals the atoms are surrounded by a ‘sea’ of free electrons. These also get heated up causing increase in the kinetic energy of the electrons, so that they also move more vigorously and further. As a result they collide with atoms in the cooler part of the metal and heat is transferred. The combination of these two processes makes conduction in metals faster.
N.B. In conduction the atoms do not move from place to place.
They simply vibrate about the same position with more energy.
Convection
This occurs in liquids and gases.
Convection is quicker than conduction but slower than radiation.
The process of convection
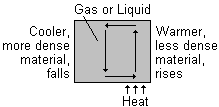
When one part of a gas or liquid is heated it expands. This means that its volume has increases and its density has decreased. Since it is now less dense than the gas or liquid above it, it rises upwards. The cooler, more dense material above it falls down and takes its place. This in turn is heated and the process repeats itself.
This results in a movement of material in a circular path.
This movement is called a convection current, and may be seen in liquids and gases.
Convection in a liquid
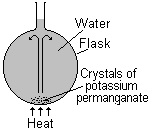
Crystals of potassium permanganate are placed in the bottom of the flask and the water is gently heated. As the warmer water rises it drags a stream of dissolving potassium permanganate with it. We therefore see a stream of purple rising. This stream then falls back down as the cooler liquid falls to replace the warmer liquid. The convection current can be followed by observing the path of the purple stream.
Convection in a gas
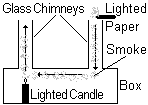
The air above the candle rises up as it is less dense than the cold air around it. Cooler air, which is denser than the warm air falls down the right hand chimney to replace it. The stream of smoke follows the convection current and smoke is pulled with the moving air across through the box where it is heated and rises out of the left hand chimney.
Examples of convection currents at work
1 Heaters – keep a room warm by setting up convection currents in the air in the room.
2 Air Conditioning – keeps room cool by setting up convection currents in the air.
3 Coastal breezes
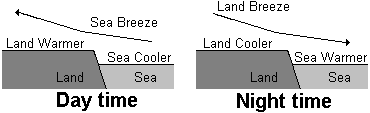
Daytime breezes - During the day the temperature of the land increases more quickly than that of the sea (due to different specific heat capacities). The hot air above the land rises and is replaced by cooler air from over the sea. A convection current is set up which causes the breeze coming in from the sea.
Night-time breezes - At night the sea has more heat to lose and cools more slowly. The air above the sea is warmer than above the land and cool air from the land sets up a breeze blows from the land to the sea.
A hot water system
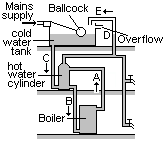
As the boiler is heated, a convection current is set up in pipe A from the top of the boiler to the top of the hot water cylinder. Cold water flows down pipe B from the bottom of the cylinder to the bottom of the boiler. This convection current allows all of the water to be heated.
When hot water is drawn off from the top of the cylinder, it is replaced by the cold water from the tank, along pipe C. Pipe E provides a pressure release overflow in case the water boils. D shows the normal water level in the overflow pipe.
Radiation
This process occurs in gases and in a vacuum. It is the quickest of the 3 methods.
It involves the movement of energy form one place to another. It is the movement of heat in the form of infra-red waves.
Good or bad absorbers of radiated heat
Heat cannot reach the tin by conduction because air is an insulator. Also hot air rises, so no convected heat reaches the tin. Radiated heat from the heater is absorbed by the two pieces of tin. This heat is then conducted through the metal. It then melts the wax and the coin falls off. The coin falls off the tin with the dull, black surface before the coin from the shiny surface. This shows that the dull, black surface absorbed more radiated heat than the shiny surface.Dull, black surfaces are better absorbers than shiny/white surfaces.A poor absorber is called a good reflector.Shiny/white surfaces are better reflectors than dull black surfaces.
Good or bad emitters of radiant heat.
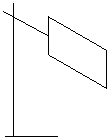
Initially the copper sheet is heated. One side is shiny and the other is dull black. If you place the backs of your hands at equal distances either side of the hot sheet, then the one opposite the dull black side feels hotter than the hand opposite the shiny side.
This shows that the dull black side emitted more radiant heat than the shiny side.
Dull, black surfaces are better emitters of radiant heat than shiny or white surfaces.
Examples of radiation
-
All hot objects give off radiant heat (infra–red radiation).
-
Heat travels from the sun through the vacuum of outer space by radiation.
-
Cooling fins on the heat exchangers on the back of refrigerators are painted black to help heat loss by radiation.
-
Tea pots and kettles are shiny so that they emit less heat by radiation.
-
Fire protection suits are shiny to reflect heat away from the fireman’s body.
People suffering from exposure / hypothermia are wrapped in shiny blankets to reflect heat back to the body and keep them warm.
