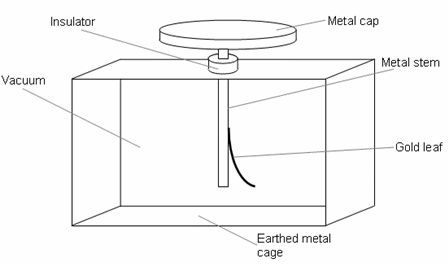
The gold leaf electroscope consists of a brass rod topped by a metal cap. A leaf of thin gold or aluminium is attached to the lower end of the rod. The rod passes through a plug of good insulating material such as alkathene, with the lower end of thee rod inside an earthed metal case with glass windows.
Uses
To detect the presence of charge on a body: if a charged body is brought close to the cap, charges of opposite polarity will be induced on the disc by being brought up from the lower end of the rod and leaf. The rod and leaf will then be charged with the same polarity charge, the same polarity as the original charged body. Rod and leaf will repel each other, and the leaf will deflect. If the body is removed the leaf will collapse, showing that the induced charge is temporary. Very small charges may be detected by this method.
To test the sign of charge on a body: a gold leaf electroscope may be negatively charged by rubbing an ebonite rod with fur and rolling the rod over the cap of the electroscope. Suppose now that a charged body is brought close to the cap. If this body is negatively charged, electrons will be repelled from the negatively charged cap down to the bottom of the rod and leaf, and the leaf will deflect further. If the body is positively charged then electrons will be attracted to it. Electrons will be drawn up from the rod and leaf so that there is less electrostatic repulsion and the deflection of the leaf will decrease.
To test the insulating properties of materials: the electroscope is charged with an ebonite rod as above. A material in someone's hand is brought into contact with the cap. If the material is a good conductor charge will flow from the cap through the material and the leaf will droop. If the material is an insulator, the deflection of the leaf will not change.
